Unlocking the door to potential therapeutic treatments for serious complications from infections
Scientists used the CLS to solve the structure of an enzyme that could help burn victims and prevent deaths from different kinds of infections.
By Sarath PeirisA surgeon operating on a patient.
Concern is growing worldwide over the increased drug resistance of two pathogens, the fungus Aspergillus fumigatus and the bacterium Pseudomonas aeruginosa, responsible for causing serious lung infections in immunocompromised people and those with chronic lung conditions such as cystic fibrosis (CF) and obstructive pulmonary disease.
Aspergillosis is the second most common hospital-acquired fungal infection, and its mortality rate is more than 50 per cent. P. aeruginosa also creates a high death toll. A major challenge facing researchers looking for new therapies to battle pulmonary infections is overcoming the defence mechanisms of these two microbes.
The pathogens collect on lung tissue, wounds, catheters, as well as heart valves, and produce slimy, sticky membranes called biofilms that attach microbes firmly to these surfaces.
The microbe colonies make a protective matrix—a sort of shell—from sugar-based polymers or protein-based polymers and DNA) to prevent the body’s immune system, or antibacterial or antifungal drugs, from penetrating. This shell makes it difficult to eradicate the pathogens that are causing infections.
For the past five years, Dr. Lynne Howell of The Hospital for Sick Children (SickKids) in Toronto and Dr. Donald Sheppard from McGill University have been working together to understand how these microbes produce their matrices, and the mechanisms that create the polymers.
“The idea is, if we understand the basic mechanisms of how the polymers are produced, we can understand ways to degrade the polymers and disrupt the matrix, making the microbes more susceptible to antibiotics,” says Howell.
Their past research has shown that a type of enzymes known as glycoside hydrolases can help break up the physical and chemical barriers of the matrices by attacking the polymers.
“We want to exploit that enzyme technology to break down the polymers, and essentially develop a therapeutic or biologic that could be used to treat infections in chronic patients,” says Howell.
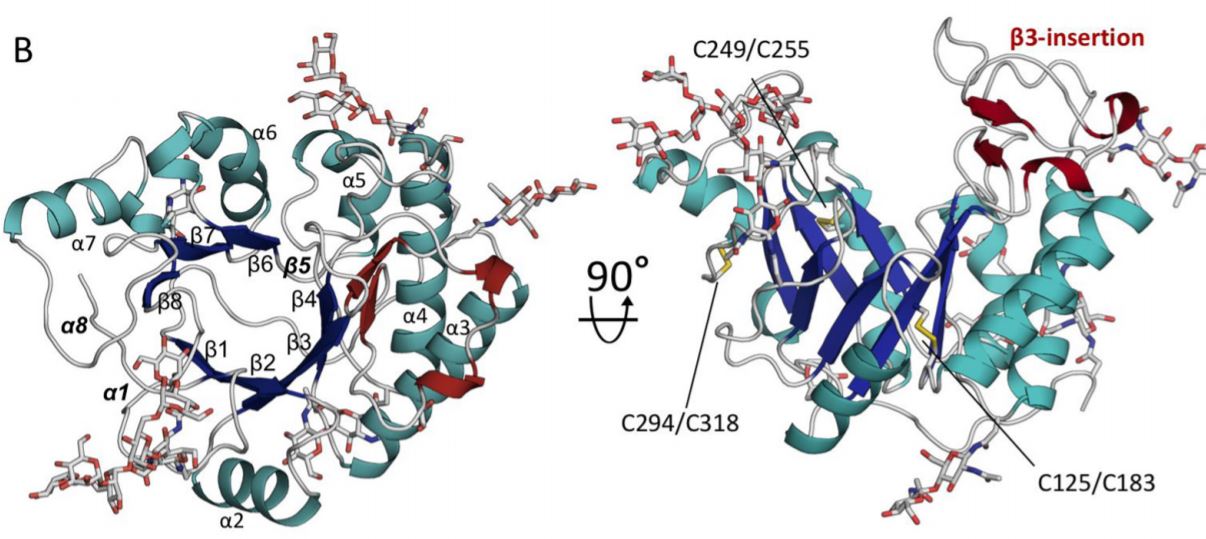
Howell and Sheppard, along with members of their teams and researchers from the Netherlands and Spain, wrote a paper recently for the Journal of Biological Chemistry about a specific enzyme called Ega3 they characterized for the first time using the Canadian Macromolecular Crystallography Facility (CMCF) at the CLS.
“The CLS was instrumental in helping us solve the structure of this enzyme and thereby understand the mechanism by which it works,” Howell says. “We were able to determine which part of the sugar polymer this enzyme works on. It’s critical information in terms of being able to potentially modify the enzyme to make it work better.”
Ega3 is involved in creating a polymer, Galactosaminogalactan (GAG), which is an integral component of the A. fumigatus matrix. Howell said the enzyme is also capable of disrupting or degrading the GAG polymer, and it can degrade polymers produced and used in P. aeruginosa matrixes.
“Our goal is to find a single enzyme that’s capable of degrading the polymers of those organisms. Then you only need one enzyme or one therapeutic,” she says.
“If we can find ways of disrupting the matrix using these enzymes, we will eventually be administering the enzyme plus an antifungal or antibiotic at the same time. What we’ve been able to show in other work is that when you add the enzyme plus a therapeutic, you enhance the activity of the antifungal to kill these matrix-encased fungi,” adds Howell. She also notes that it could take at least ten years before a therapy could be developed, tested and brought into the marketplace.
Characterizing the Ega3 enzyme has also enabled researchers to investigate the similarity in the chemical structure of polymers produced by other organisms, Howell said, opening the door to potential therapeutic treatments for different microbial infections, such as burn wounds and other wounds that are difficult to heal.
Howell is a Senior Scientist in Molecular Medicine at SickKids, a Professor at the University of Toronto’s Biochemistry Department, and a member of the CLS Board of Directors. Sheppard is also Director, McGill Interdisciplinary Initiative in Infection and Immunity, Professor and Chair of the Department of Microbiology & Immunology, and Professor in the Department of Medicine at McGill University.
Bamford, Natalie C., François Le Mauff, Adithya S. Subramanian, Patrick Yip, Claudia Millán, Yongzhen Zhang, Caitlin Zacharias et al. "Ega3 from the fungal pathogen Aspergillus fumigatus is an endo-α-1, 4-galactosaminidase that disrupts microbial biofilms." Journal of Biological Chemistry 294, no. 37 (2019): 13833-13849. DOI: 10.1074/jbc.RA119.009910.
For more information, contact:
Victoria Schramm
Communications Coordinator
Canadian Light Source
306-657-3516
victoria.schramm@lightsource.ca
